
Tandem Pd(II)-Catalyzed Vinyl Ether Exchange−Claisen Rearrangement as a Facile Approach to γ,δ-Unsaturated Aldehydes | The Journal of Organic Chemistry
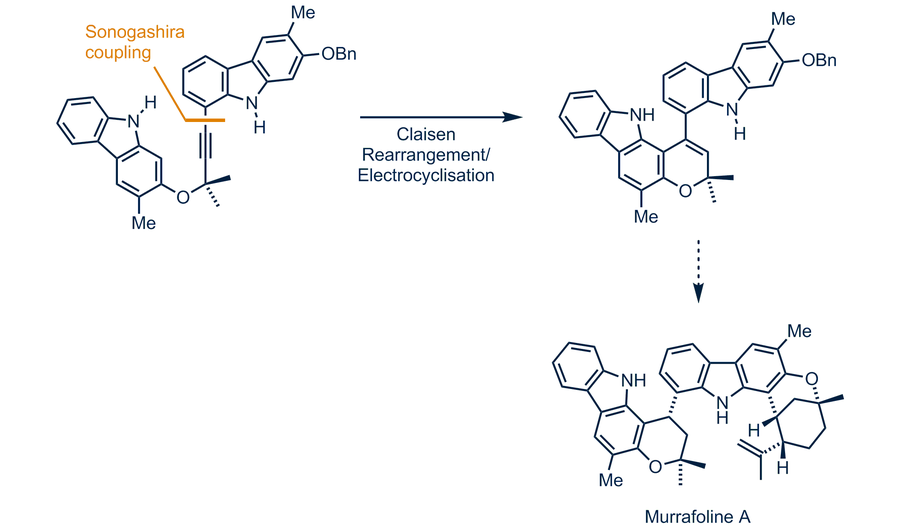
Palladium(II)-catalyzed synthesis of carbazole alkaloids — Chair of Organic Chemistry II — TU Dresden
![Divergent Reactivity in Pd-Catalyzed [3,3]-Sigmatropic Rearrangement of Allyloxy- and Propargyloxyindoles Revealed by Computation and Experiment | Organic Letters Divergent Reactivity in Pd-Catalyzed [3,3]-Sigmatropic Rearrangement of Allyloxy- and Propargyloxyindoles Revealed by Computation and Experiment | Organic Letters](https://pubs.acs.org/cms/10.1021/acs.orglett.8b02864/asset/images/medium/ol-2018-02864n_0004.gif)
Divergent Reactivity in Pd-Catalyzed [3,3]-Sigmatropic Rearrangement of Allyloxy- and Propargyloxyindoles Revealed by Computation and Experiment | Organic Letters

Copper(II)- and Palladium(II)-Catalyzed Enantioselective Claisen Rearrangement of Allyloxy- and Propargyloxy-Indoles to Quaternary Oxindoles and Spirocyclic Lactones | The Journal of Organic Chemistry

Pd-Catalyzed Rearrangement of N-Alloc-N-allyl Ynamides via Auto-Tandem Catalysis: Evidence for Reversible C–N Activation and Pd(0)-Accelerated Ketenimine Aza-Claisen Rearrangement | Organic Letters

Scheme 22. Pd-catalyzed rearrangement of N-alloc-N-allyl ynamides via... | Download Scientific Diagram

Palladium‐Catalyzed Tunable Functionalization of Allylic Imidates: Regioselective Aminodiacetoxylation and Aziridination - Cui - 2011 - Angewandte Chemie International Edition - Wiley Online Library

The Retro‐Claisen Rearrangement of 2‐Vinylcyclopropylcarbonyl Substrates and the Question of its Synthetic Potential - Zhang - 2020 - European Journal of Organic Chemistry - Wiley Online Library
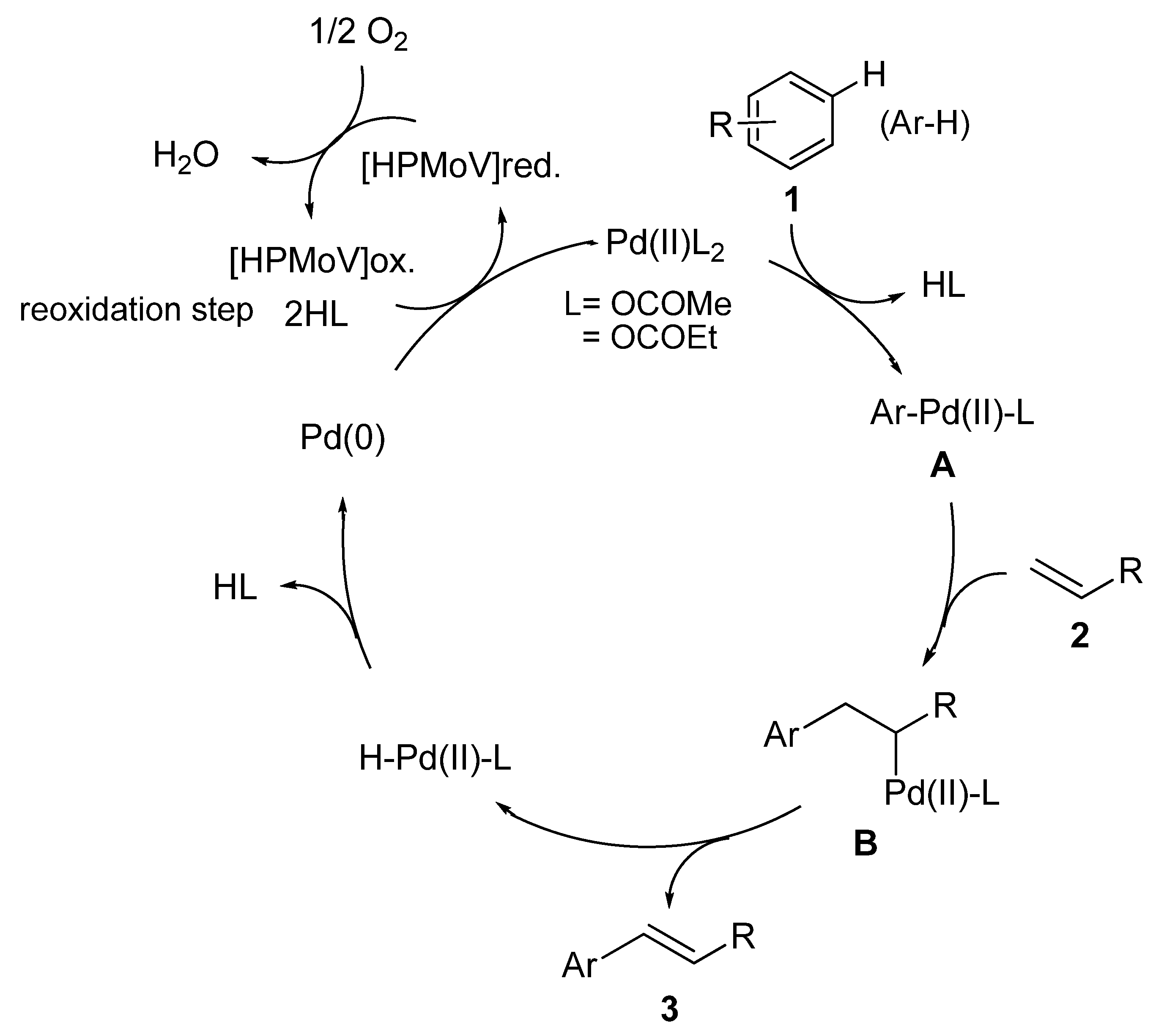
Molecules | Free Full-Text | Pd(II)/HPMoV-Catalyzed Direct Oxidative Coupling Reaction of Benzenes with Olefins
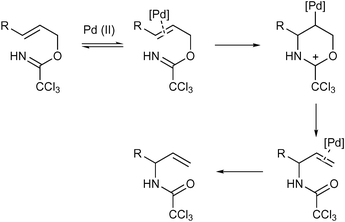
A highly stereoselective ether directed palladium catalysed aza-Claisen rearrangement - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/B501346C
Palladium(II)-catalyzed synthesis of carbazole alkaloids — Chair of Organic Chemistry II — TU Dresden

An Enantioselective Claisen Rearrangement Catalyzed by N-Heterocyclic Carbenes | Journal of the American Chemical Society

Total Synthesis of (+)‐Cinereain and (−)‐Janoxepin through a Fragment Coupling/Retro‐Claisen Rearrangement Cascade - Ronzon - 2022 - Angewandte Chemie International Edition - Wiley Online Library

Tandem Pd(II)-Catalyzed Vinyl Ether Exchange−Claisen Rearrangement as a Facile Approach to γ,δ-Unsaturated Aldehydes | The Journal of Organic Chemistry

The proposed mechanisms for the Claisen rearrangement of aryl propargyl... | Download Scientific Diagram
![Palladium-catalyzed sequential reaction via Sonogashira coupling, isomerization, Claisen rearrangement and [4 + 2] cycloaddition sequence for the rapi ... - RSC Advances (RSC Publishing) DOI:10.1039/C1RA00452B Palladium-catalyzed sequential reaction via Sonogashira coupling, isomerization, Claisen rearrangement and [4 + 2] cycloaddition sequence for the rapi ... - RSC Advances (RSC Publishing) DOI:10.1039/C1RA00452B](https://pubs.rsc.org/image/article/2012/RA/c1ra00452b/c1ra00452b-s1.gif)
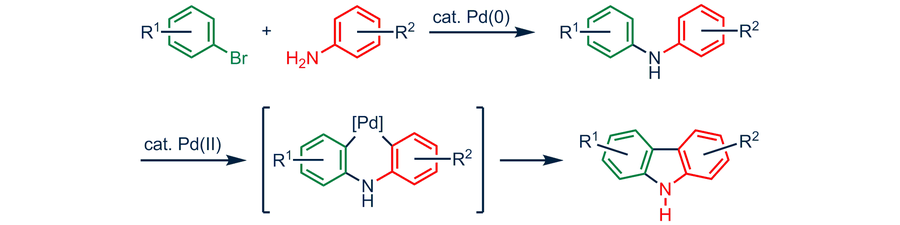
Palladium-catalyzed sequential reaction via Sonogashira coupling, isomerization, Claisen rearrangement and [4 + 2] cycloaddition sequence for the rapi ... - RSC Advances (RSC Publishing) DOI:10.1039/C1RA00452B
![PDF) A new palladium(II)-catalyzed [3,3] aza-Claisen rearrangement of 3-allyloxy-5-aryl-1,2,4-oxadiazoles | Antonio Piccionello - Academia.edu PDF) A new palladium(II)-catalyzed [3,3] aza-Claisen rearrangement of 3-allyloxy-5-aryl-1,2,4-oxadiazoles | Antonio Piccionello - Academia.edu](https://0.academia-photos.com/attachment_thumbnails/44982140/mini_magick20190213-14539-zxuyqy.png?1550072610)
PDF) A new palladium(II)-catalyzed [3,3] aza-Claisen rearrangement of 3-allyloxy-5-aryl-1,2,4-oxadiazoles | Antonio Piccionello - Academia.edu

Toward a symphony of reactivity: cascades involving catalysis and sigmatropic rearrangements. - Abstract - Europe PMC




![Catalysts | Free Full-Text | Recent Advances in Catalytic [3,3]-Sigmatropic Rearrangements Catalysts | Free Full-Text | Recent Advances in Catalytic [3,3]-Sigmatropic Rearrangements](https://www.mdpi.com/catalysts/catalysts-12-00227/article_deploy/html/images/catalysts-12-00227-g001.png)